Neurological conditions claim the lives of thousands of children every year. New treatments in the womb may save them
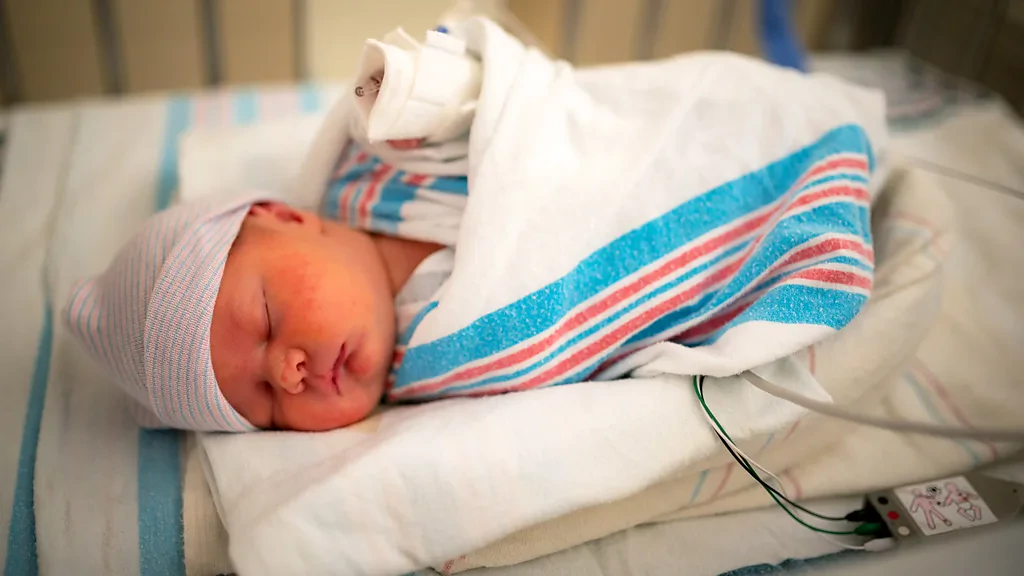
Cutting-edge therapies delivering treatment to foetuses diagnosed with neurological defects have the potential to change natal care as we know it.
Michelle Johnson’s fourth pregnancy was all going according to plan until the specialist giving her the standard 20-week ultrasound stiffened slightly while prodding her belly. The ultrasonographer got "really awkward" and was erratically asking questions, Johnson recalls, like if they already had kids. Several days later, the department’s head radiologist called. His tone was sombre as he fumbled for words and said that it was rare in his experience, but Johnson needed to see a gestational foetal medicine specialist immediately because her child had spina bifida.
Known to science as myelomeningocele, spina bifida is when the spinal cord – which starts developing in children as a cannoli-shaped tube, folding onto itself to encapsulate the nervous system – doesn't fully fuse close. It leaves the superhighway of nerves to spill into a small bulge somewhere along the spine. This nervous system birth defect can lead to lifelong cognitive issues as well as chronically impaired mobility and paralysis from the hips down.
"It was just devastating," says Johnson, who was 35 years old at the time and living in Portland, Oregon, US. "I was just in shock."
Each year around 1,400 babies are born in the US with spina bifida. The exact cause of this complex condition is not known, but it is thought to involve a combination of genetic and environmental factors. Low levels of folic acid intake during pregnancy or certain anti-seizure medications, for example, have been associated with a higher risk of the condition, but it is not clear how great a role these play.
Spina bifida is customarily treated in the 24 to 48 hours immediately following childbirth: surgeons sew up the spinal cord and tuck it back into the baby's body, preventing the condition from degenerating any further. But while making calls for appointments with a specialist, a nurse on the phone told Johnson about a new programme in California that uses stem cells to treat children with spina bifida while they are still in the womb.
If she chose to be involved, her baby would be the second human patient to ever undergo this type of treatment. Johnson knew it was a chance she wanted to give her unborn child.
Since the procedure had to happen before the 26th week of pregnancy, "it was like a race", says Johnson. Several scans, blood tests, and interviews later, she embarked on something larger than life.
Prenatal screening for neurological conditions has progressed in leaps and bounds over the past couple of decades. Technology including genetic analysis, neuroimaging, and high-resolution foetal magnetic resonance imaging (MRI) are allowing doctors to peer into the nervous systems of developing foetuses and already diagnose them – earlier and more frequently – with any life-altering conditions they'll experience once born. But throughout this advancement, there hasn't been much doctors could do about those diagnoses until the child emerged from the womb. And a significant portion of crucial brain development happens long before a child is born.
Now, a new wave of pioneering in-utero neuroscience therapies are helping to change that. Several seminal trials are underway to test both surgical and medical treatments allowing doctors to reverse conditions in babies before they are born. And the field is "right on the precipice" of a whole new dimension of therapies, says Jeffrey Russ, a pediatric neurologist at Duke University who recently wrote an academic essay describing in-utero treatment as the "next frontier" in neurology.
One of these frontier treatments is the current first Food and Drug Administration (FDA) approved clinical trial treating spina bifida in utero with placental stem cells. The project, known as the CuRe Trial, is the culmination of 25 years of work of foetal surgeon Diana Lee Farmer, from the University of California, Davis.
In-utero operations where the spinal cord opening is sewn shut have become standard care for cases of very severe spina bifida. They slow the degeneration of the disease throughout the pregnancy and improve patient outcomes more than post-birth surgeries, as years of work by Farmer suggest. But her new project aims to take it a step further. By mending the open neural tube with a patch seeded with stem cells from bits of the mother's placenta – known as "mesenchymal stromal cells", derived and cultured in a painstakingly precise four-day process – the cells should actively go in and reverse the damage that has already occurred by the time of diagnosis.
These stem cells "are very smart", says Aijun Wang, the bioengineer who developed this stem cell technology for the CuRe trial. "They can protect neurons from being killed by the environment."
Preliminary data from running this experiment on lambs with spina bifida suggested the treatment allowed them to prance around without any noticeable disability when they would have otherwise been paralysed in their hind legs. The same was observed when the procedure was performed on bulldogs.
More than 30 people were in the operation room when Johnson, just a day shy of the 26-week pregnancy cutoff, became the second human patient to undergo this surgery. An incision was made into her watermelon-shaped belly, the uterus was pulled almost fully out of the body, and the foetus was floated up to the opening in the uterus. This allowed doctors to reach the hernia on the minuscule baby spine, and delicately apply the stem cell patch. The doctors used special microscopes to perform their surgery because the baby was so small.
If the child – a boy to be named Tobias – had remained untreated, he would have been born with paralysis from the hips down. On 1 February 2022, he was born via caesarian-section at 7lbs 13oz (3.5kg), legs kicking, toes wriggling. "It just feels like we won the lottery," says Johnson.
Tobias will have to be monitored until he turns 30 months old – his last in-person visit is upcoming this summer – before officially assessing the full safety and efficacy of the procedure, for the sake of the experiment. And doctors will likely continue to follow him until he turns at least five. At the time of writing, 10 more patients have received CuRe treatment, and Farmer's team has secured $15m (£12m) of funding for 29 more patients, with the hopes of enrolling about 10 patients a year. It will not be till 2028 that Farmer’s team will be able to review all of the data collected and confirm whether this new therapy could become standard for children across the country.
"I'm hopeful that we may be able to make a very significant improvement in the outcome of these kids with spina bifida," says Farmer. "But like every good scientific project, you answer one question and that opens the door to another question."
This is cutting-edge science for anatomic conditions that can be surgically repaired, according to Russ. But another frontier where in-utero therapies have the potential to turn the tables of neurological conditions in newborn babies is where therapies can be delivered at a molecular or genetic level. This is a "totally new concept" that will "open a whole new realm", says Russ.
His colleagues at Duke University contributed to the design of a protocol for the first-ever in-utero treatment for Pompe's disease – a rare genetic disease that causes cells to hold onto too many complex sugars, leading to neurological conditions, breathing problems, heart issues, and muscle weakness. Most patients die within one or two years of birth.
Since Pompe's is caused by the lack of an enzyme called acid alpha-glucosidase, it's usually treated with enzyme replacement therapy (ERT), where children receive regular injections of the enzyme. Like with spina bifida, though, data has shown that starting ERT as soon as the baby is born can improve symptoms but it cannot fully stop the disease from manifesting.
So, when doctors at The Ottawa Hospital in Ontario, Canada, ran tests on an unborn girl called Ayla Bashir in February 2021, revealing she had inherited the same genes that led two of her siblings, Zara and Sara, to be diagnosed with Pompe's after birth – they knew they had to act quickly. Both Zara and Sara had died aged 29 months and eight months, respectively. But with Ayla's in-utero diagnosis, the medical team could intervene sooner.
On March 24, 2021, doctors delivered the first dose of enzyme replacement to Ayla while she was still in her mother's womb at 24 weeks and five days of gestation. They injected into the umbilical vein a liquid formula containing a copy of the missing enzyme called alglucosidase alfa. This approach allows the manufactured enzyme to make its way into the foetus's bloodstream while it is still developing. It means it barely notices the drug as foreign to itself, and doesn't have the strong immune response that can occur during treatment after birth. Six more infusions every two weeks followed. Ayla was born on June 22, 2021, and ever since she has been getting enzyme injections every week.
Similarly to Tobias, doctors will continue to follow Ayla for at least five years to monitor any disease progression, as the therapy does not completely prevent irreversible organ damage. But Ayla's story paves the way for setting up prenatal medical treatments that intervene in disorders like these with a simple injection.
"We were hoping to change the paradigm for when you can treat a genetic disease," says Tippi Mackenzie, one of the foetal surgeons at the University of California, San Francisco who led the development of the protocol which was used to treat Ayla.
There are several treatments currently offered to newborns that could potentially be offered during the foetal phase, says Mackenzie. She has established an ongoing five-year clinical trial for a total of 10 patients underway in California to help officially establish in-utero ERT as an approved procedure for Pompe's and other rare diseases such as Neuronopathic Gaucher disease, Mucopolysaccharidosis, and Wolman disease. Two babies with Mucopolysaccharidosis were already treated as part of the trial and "updates are positive", says Mackenzie. They are continuing to enroll patients for the trial, which is open for international patients.
Developing a foetal treatment for conditions like these would also help to raise awareness about the need for more testing for genetic diseases in the first place – "changing the equation" by allowing a virtuous cycle of more diagnoses and more treatments, says Mackenzie. "The diagnosis and treatment, I call them a yin and yang, they go together," she says.
And while enzyme replacement is the least invasive type of treatment for genetic disorders – and requires multiple dosages throughout a patient's lifetime – this new method could be adapted to deliver other hotly debated gene therapies for editing an unborn baby's DNA, by either snipping out a defective gene or replacing a missing one.
Mackenzie's work is "laying the foundation for these types of advanced therapies in the future", says William Peranteau, a professor of surgery at the Children's Hospital of Philadelphia. "If those trials can demonstrate a benefit to treating the diseases before birth with an enzyme replacement therapy, then the obvious next question is a more definitive therapy like in-utero gene editing."
In a series of groundbreaking experiments, Peranteau has used the genome-editing technique Crispr to tweak the genetic code of a few mice still in the womb to treat them for a genetic skin condition, a genetic lung disease, and a genetic metabolic disorder affecting the rodents' liver.
When it comes to estimating how long it'll be until gene-editing therapies will get human trials, though, it's always a very difficult question to answer, and "it always takes longer than we want or expect it to", says Peranteau. Maybe five to 10 years. "It's a matter of just doing the work," he says.
For now, as these trials pick up pace, it'll be imperative to consider the ethical and practical implications of these advancements.
"We'll have to start with really specific examples, where it's very clear that the benefits outweigh the risks," says Russ. Obviously, not all conditions can and should be treated with stem cells, enzyme replacement, and gene-editing technology before birth.
And it's still too early to have a clear picture of the long-term effects of these in-utero treatments. Most of the patients from these in-utero trials are still babies or very young children. We also still don't have long-term data from adult patients who are currently undergoing gene editing therapies, either. So while surgeries and chemical therapies like those developed by Farmer and Mackenzie are mostly short-lived procedures, once doctors edit the genetic code of an unborn baby these changes, and their effects, will be forever.
Crucially, in-utero therapies are unique procedures with double the risks and stakes, since they involve the mother as well as her unborn child, says Russ: "You're not just treating one patient, you're treating two."
Johnson’s family travelled back to California a little over a year after the procedure. Johnson got to meet other mothers from the CuRe trial, and the hospital staff all took turns coming by, saying hello, and playing with Tobias in the courtyard, bringing him cake and candles to celebrate his first birthday.
"It was really special," says Johnson. "Really full circle for them to see all the work they're doing and to meet this miracle baby and see how healthy and happy he is."
At the time of writing, Tobias is more than two years old, and he has learned to walk.
-BBC


Leave Comment